Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ota, Masakazu; Kwamena, N.-O. A.*; Mihok, S.*; Korolevych, V.*
Journal of Environmental Radioactivity, 178-179, p.212 - 231, 2017/11
Times Cited Count:14 Percentile:43.09(Environmental Sciences)Environmental transfer models assume that organically-bound tritium (OBT) is formed directly from tissue-free water tritium (TFWT) in environmental compartments. Nevertheless, studies in the literature have shown that measured OBT/TFWT ratios are variable. The importance of soil-to-leaf HTO transfer pathway in controlling the leaf tritium dynamics is not well understood. A model inter-comparison of two tritium transfer models (CTEM-CLASS-TT and SOLVEG-II) was carried out with measured environmental samples from an experimental garden plot set up next to a tritium-processing facility. The garden plot received one of three different irrigation treatments - no external irrigation, irrigation with low tritium water and irrigation with high tritium water. The contrast between the results obtained with the different irrigation treatments provided insights into the impact of soil-to-leaf HTO transfer on the leaf tritium dynamics. Concentrations of TFWT and OBT in the garden plots that were not irrigated or irrigated with low tritium water were variable, responding to the arrival of the HTO-plume from the tritium-processing facility. In contrast, for the plants irrigated with high tritium water, the TFWT concentration remained elevated due to a continuous source of high HTO in the soil. Calculated concentrations of OBT in the leaves showed an initial increase followed by quasi-equilibration with the TFWT concentration. In this quasi-equilibrium state, concentrations of OBT remained elevated and unchanged despite the arrivals of the plume. These results from the model inter-comparison demonstrate that soil-to-leaf HTO transfer significantly affects OBT/TFWT ratio in the leaf regardless of the atmospheric HTO concentration, only if there is elevated HTO concentrations in the soil. The results of this work indicate that assessment models should be refined to consider the importance of soil-to-leaf HTO transfer to ensure that dose estimates are accurate and conservative.
Irisawa, Keita; Meguro, Yoshihiro
Journal of Nuclear Science and Technology, 54(3), p.365 - 372, 2017/03
Times Cited Count:2 Percentile:19.37(Nuclear Science & Technology)Kozai, Naofumi; Onuki, Toshihiko; Komarneni, S.*
Journal of Materials Chemistry, 17(12), p.2993 - 2996, 2002/12
Here we report the extremely high and selective uptake of selenium oxyanions by a novel exchanger, Ni Zn
Zn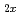 (OH)
(OH) (OCOCH
(OCOCH )
)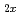 ・nH
・nH O that has brucite-type hydroxide layers with OCOCH
O that has brucite-type hydroxide layers with OCOCH anions in the interlayers. This Ni-Zn basic salt exhibited very high selectivity for Se(IV) (Kd = 9.0x10
anions in the interlayers. This Ni-Zn basic salt exhibited very high selectivity for Se(IV) (Kd = 9.0x10 ml/g with an initial Se(IV) concentration of 1x10
ml/g with an initial Se(IV) concentration of 1x10 M) in the presence of 0.1M Cl
M) in the presence of 0.1M Cl solution while the well known anionic clay, Mg
solution while the well known anionic clay, Mg Al(OH)
Al(OH) NO
NO ・nH
・nH O showed a Kd of 6.0x10
O showed a Kd of 6.0x10 ml/g under the same conditions. The uptake of Se(IV) on the Ni-Zn basic salt was found to be irreversible when treated with solutions containing 1N Cl
ml/g under the same conditions. The uptake of Se(IV) on the Ni-Zn basic salt was found to be irreversible when treated with solutions containing 1N Cl , 1N NO
, 1N NO
 , or 1N PO
, or 1N PO
 , while the Se(IV) sorbed on anionic clay was easily desorbed in a 1M Cl
, while the Se(IV) sorbed on anionic clay was easily desorbed in a 1M Cl solution. This novel exchanger also showed high Kd (2.6
solution. This novel exchanger also showed high Kd (2.6 10
10 ml/g at an initial Se concentration of 1
ml/g at an initial Se concentration of 1 10
10 M) for Se(VI) and therefore it is expected to be useful for decontamination and removal of selenium oxyanions from contaminated water.
M) for Se(VI) and therefore it is expected to be useful for decontamination and removal of selenium oxyanions from contaminated water.
Furukawa, Jun*; Nakanishi, Tomoko*; Matsubayashi, Masahito
Nuclear Instruments and Methods in Physics Research A, 424(1), p.116 - 121, 1999/00
Times Cited Count:19 Percentile:78.49(Instruments & Instrumentation)no abstracts in English
E.A.Hegazy*; ; A.Rabie*; A.M.Dessouki*;
J.Appl.Polym.Sci., 26, p.3871 - 3883, 1981/00
Times Cited Count:56 Percentile:92.3(Polymer Science)no abstracts in English